
Sometimes you just have to get silly with some clip art…
Best wishes for a wonderful 2016!
Eastern Michigan University

Sometimes you just have to get silly with some clip art…
Best wishes for a wonderful 2016!
My terrific collaborators at Herpetological Resource and Management put together this map of the Mudpuppy samples we have acquired so far from the Huron-Erie Corridor (St. Clair-Detroit River System) here in SE Michigan. Each yellow dot represents a capture record, for which we have a tissue sample. We never could have done this on our own, and owe a huge debt of gratitude to biologists at the U.S. Fish and Wildlife Service, U.S. Geological Survey, Michigan Department of Natural Resources, and Belle Isle Aquarium for their assistance!
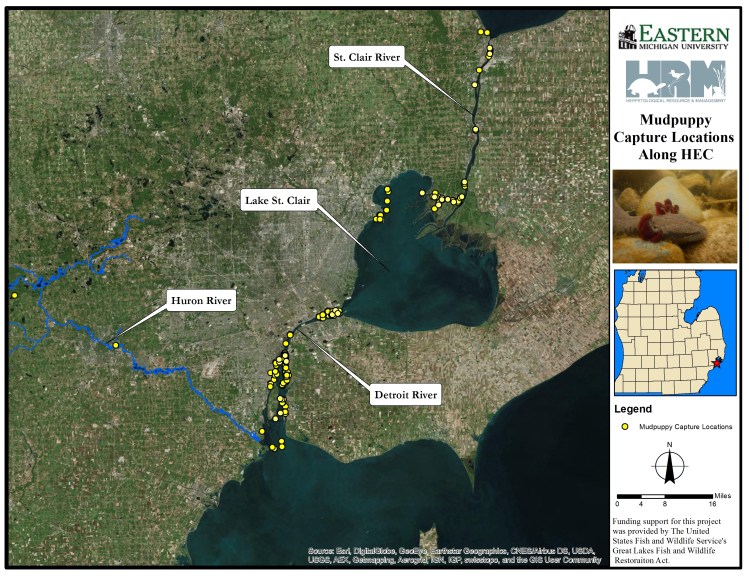 This is going to be an amazing dataset to look at gene flow among the lake and river systems, and to see whether things like surrounding landscape (and contaminant input) may be fragmenting populations. We also have a set of historic samples from the Detroit River, so we can see how effective population size has changed over time.
This is going to be an amazing dataset to look at gene flow among the lake and river systems, and to see whether things like surrounding landscape (and contaminant input) may be fragmenting populations. We also have a set of historic samples from the Detroit River, so we can see how effective population size has changed over time.
We’ve also made some terrific progress on the restoration side of the project, placing these structures at a number of sites to improve Mudpuppy habitat. They seem to work just as well in real life as they do in the adorable schematic drawing below. In fact, we’ve found Mudpuppies using these structures in as little as 48 hours after placement. Very exciting to be learning more about how to conserve these remarkable animals!
(Photos courtesy of HRM; artwork by Tricia Brockman)

There was a neat event yesterday here at EMU – the State of the Strait, a biennial conference with the goal of coordinating conservation action along the St. Clair-Detroit River System (a.k.a. SCDRS, a.k.a. the Huron-Erie Corridor, a.k.a. the focal region for our ongoing Mudpuppy work). I attended part of the meeting, in between teaching two classes. My collaborator on the Mudpuppy project, Dave Mifsud, was able to stay for the entire event. There were some terrific talks and a lot of extremely valuable networking – thanks to the organizers for putting together such a terrific meeting! It is really exciting to know there are so many organizations and agencies collaborating for the improvement of our local environment.
We presented a poster that is a broad overview of the Mudpuppy project, describing the goals of both the restoration and research efforts. Here is the pdf if you’d like to take a look!
State of the Strait_Mudpuppy Project

Sometimes in science you start a project thinking it might be a SMALL project… single focal species, limited geographic area, maybe a year or two of fieldwork, and a nice thesis for a Master’s student. Next thing you know, your freezer is full of hundreds of samples from seven states and two provinces, and you have three count ’em three Master’s students working on different aspects of this “small project.”
Am I complaining? Heck no! This is awesome, and it’s exactly what happened with the Mudpuppy (Necturus maculosus) study that Dave Mifsud and I are collaborating on. Broadly, we had three goals when we started the project.
All of this work was to be conducted within the Huron-Erie Corridor here in Southeast Michigan, and the project was funded by the Great Lakes Fish and Wildlife Restoration Act. The proposal included a ton of amazing collaborators, including folks from the Belle Isle Conservancy, Michigan Department of Natural Resources, U.S. Geological Survey, U.S. Fish and Wildlife Service, and the Michigan Sea Grant. We already had a fantastic team (GO TEAM MUDPUPPY), and from there the project just seemed to take on a life of its own.
I mean, how can you not love these guys???
We found out-of-state collaborators, and out-of-state collaborators found us. Turns out a lot of biologists across the Midwest are concerned about this species. There is lots of anecdotal evidence suggesting Mudpuppies are in decline throughout the region, but there is not a heck of a lot of good data. We’re hoping we can help change that!
The type of population genetic analysis we’re conducting can be really valuable in informing conservation actions. First, we can figure out if populations are highly isolated and therefore more likely to die out (this seems likely, given that Mudpuppies are fully aquatic and their river homes are frequently fragmented by dams). Second, we can estimate effective population size, and determine if populations have recently undergone major reductions. This would be especially informative in areas that are suspected to have lost large numbers of Mudpuppies due to things like pollution, sedimentation, or overexploitation.
We’re currently in the depths of processing samples, so I can’t report much in the way of results quite yet. My grad student Amber Stedman has been working on the large-scale phylogeography of the species, and so far we seem to be finding a deep division between eastern and western populations from the Great Lakes region. We’re going to end up with an astounding number of samples from Minnesota, due to the efforts of Krista Larson of the Minnesota DNR. Krista wins my award for “Badass Biologist of the Year”… this is how you sample mudpuppies in Minnesota.
All pics above by Krista Larson, Badass Biologist of the Year
I am really excited for what we might learn from these samples. Krista has collected such a large number of samples, and with such great spatial coverage, that we should be able to delineate which populations are at greatest risk of local extinction. That in turn will hopefully help Minnesota biologists determine the best management practices for this species.
This is the research “sweet spot” for me. With amphibians in decline globally, I want to make sure that, as much as possible, the research done in my lab is directly informing conservation actions. Projects like this Mudpuppy work are golden opportunities to collaborate with people both locally and regionally, and hopefully make “real world” contributions to the conservation of this remarkable species.
This has been a fun week for my lab. My second Master’s student, Patrick Terry, successfully defended his thesis on Tuesday. Patrick came to the lab from Ohio University, where he had worked on some turtle projects and developed a strong interest in all things Testudines. Here at EMU, Patrick has been an incredibly independent and self-motivated student, and basically took on developing (and funding!) his own project. His thesis addresses a long-standing question among Michigan herpetologists: is the red-eared slider native or introduced in the state? The mystery arises from the fact that we’re at the edge of the native range. There are some other herps with similarly odd, disjunct populations in the state that are suspected to result from glacial refugia or postglacial recolonization. There is also some (somewhat contentious) fossil evidence indicating that the turtles may have been present in the state long ago. In short, it’s a question that genetic evidence might help shed some light on.
Patrick did a lot of sampling on his own, and worked with collaborators in other states to collect tissue samples for genetic analysis. In the end, he had sufficient sample size from three populations in Michigan, two in Indiana, and one in Ohio.
 This was a nice set for comparison, because the Indiana sites were within the native range of the turtle (purple in the map above), as it is currently understood. The Ohio population is well outside the range and considered introduced.
This was a nice set for comparison, because the Indiana sites were within the native range of the turtle (purple in the map above), as it is currently understood. The Ohio population is well outside the range and considered introduced.
The pictures above show Patrick after flipping his kayak to catch a turtle (L), and taking a tissue sample for genetic analysis (R). The joys of fieldwork! 🙂
Patrick conducted a whole suite of analyses to look at how his populations compared to one another, and they all had the same answer: one of his Michigan sites is very different from the rest of the sites, which are all quite similar to one another. The picture below is a visualization from the program Geneland.
 This is basically a “heat map” showing how likely it is that various sampled sites cluster together. You can see that most sites are quite similar (all the red-orangy-ness). But there is one, TL, which is “white-hot-different.” All of the results reflected this same pattern. Among the sites Patrick sampled, it looks like that one is a likely introduction (and probably from a fairly distant source population). The other Michigan sites are more ambiguous. They may be naturally occurring, or they may be introduced from more regional (genetically similar) source populations.
This is basically a “heat map” showing how likely it is that various sampled sites cluster together. You can see that most sites are quite similar (all the red-orangy-ness). But there is one, TL, which is “white-hot-different.” All of the results reflected this same pattern. Among the sites Patrick sampled, it looks like that one is a likely introduction (and probably from a fairly distant source population). The other Michigan sites are more ambiguous. They may be naturally occurring, or they may be introduced from more regional (genetically similar) source populations.
Patrick is preparing a manuscript for submission, so keep an eye out for the full report soon! In the meantime, congrats to Patrick on the terrific work and the shiny new degree!
The lab had a prospective student visiting yesterday, Jeff Bartman (currently working with Dr. Jennifer Moore at GVSU). It was a great excuse for a few of us to get out to the George Reserve for a fall visit. We’d had a very rainy day on Wednesday, courtesy of the remnants of Hurricane Patricia, so I was optimistic that we might see some salamanders despite the cool temperatures. And indeed, one of the first boards we flipped had a salamander underneath. Given that we have about a 9:1 ratio of unisexual Ambystoma to “real” blue-spotted salamanders at this site, this pretty girl is probably a uni. Since it was our first salamander of the day, there were many photo ops (top to bottom: Jenny Sutherland, Amber Stedman, and Jeff Bartman).


 We didn’t need to worry, though – there were piles more salamanders to be found! Sometimes literally. The next board I flipped had this cute little juvenile, likely one of this year’s metamorphs.
We didn’t need to worry, though – there were piles more salamanders to be found! Sometimes literally. The next board I flipped had this cute little juvenile, likely one of this year’s metamorphs.
And then we had quite a number of “jackpot boards”!
The only other amphibians we came across were a few very chilly Western Chorus Frogs (Pseudacris triseriata). There is one in the photo above, but here’s an individual better illustrating the three stripes from which they get their name.
All in all, a very fun day in the field. Thanks to Amber, Jenny and Jeff for joining me!
As I posted about recently, lab undergraduate researcher Marisa Hildebrandt attended the Sigma Xi Student Research Conference this past weekend. Not only did she report having a terrific time, but the following news came to my phone on Saturday evening:
 She also sent a photo of the swanky medal she gets to wear around town now.
She also sent a photo of the swanky medal she gets to wear around town now.
 In case you’d like to learn more about her project, here is a pdf of her award-winning poster.
In case you’d like to learn more about her project, here is a pdf of her award-winning poster.
In all seriousness, Marisa is an incredibly hard worker and has been a terrific asset to the lab during her years here. We will sure miss her when she graduates. I couldn’t be prouder of her initiative to attend and present at this conference, and her great success in doing so. Congratulations, Marisa!!
If you are part of the online scientific blogging and/or tweeting community, you have probably heard of #upgoerfive. This was an exercise inspired by an xkcd illustration of a rocket, labeled using only the 1000 most common words in the English language. Scientists challenged each other to describe research programs or scientific concepts in the same way (there’s even a handy text editor!). I heard about it over on Dynamic Ecology, and had to give it a shot.
 My #upgoerfive attempt at explaining my research system
My #upgoerfive attempt at explaining my research system
OK, so that’s pretty silly, and I had fun doing it. But, is it actually a useful exercise in scientific communication? There is a great discussion of this question in the comments of the Dynamic Ecology post I linked above. I do see the entertainment value in this exercise, but truly, I could probably use the word “salamander” and most folks would know what I was talking about (and if not, I would offer to get them out in the woods! STAT!).
 THIS IS A SALAMANDER. IT IS BEAUTIFUL.
THIS IS A SALAMANDER. IT IS BEAUTIFUL.
So, inspired by thinking about all of this (and by having this brand new blogging platform!), I decided to attempt to “level up” my explanation above. My imaginary audience is a group of table-mates at a dinner party. [Editor’s note: Table-mates who are exceedingly tolerant of what turned out to be a rather long explanation.] They are smart people, but they’re not biologists. They may not even know we have salamanders in Michigan (THE HORROR). If I start throwing around terms like “tetraploidy” and “kleptogenesis,” they may suddenly find themselves “having to go make a call, it was so nice chatting, take care hmm?”
I’m going to divide this exercise into two parts, since my research falls into two overlapping but distinct areas. Part 1 (you’re reading it!) will focus on the all-female salamanders that I described above, while Part 2 will describe more applied, conservation-related work (I’ll link it here once it’s posted). I’d love feedback on this exercise, especially from my non-biologist friends and family. Have I managed to avoid jargon? Will you hang out with me at the dinner party?
 Don’t make me drink this beer alone
Don’t make me drink this beer alone
I’d also love to read about what other people are up to… tweet it up with #DinnertimeSciComm. And if you’re in the U.K., get me some of that beer please and thank you. 🙂
Why do we care about these crazy all-female salamanders???
Everyone is interested in sex, and evolutionary biologists are no exception. The fact that most animals reproduce sexually is actually something of an evolutionary mystery. Animals that can reproduce asexually – think cloning – should actually have a numerical advantage. With cloning, every individual can produce offspring directly (rather than requiring a male and a female to make a kid). Plus, a clone-mom gets to pass on 100% of her genes, whereas a sexual reproducer only passes on half! Passing on genes is the name of the game in evolution… so why aren’t clonal animals more common?
One possible explanation is that populations of clones lack genetic variation. If the environment changes in some way, those clonal animals are more likely to go extinct. If a new disease wipes one of them out, it’s going to wipe all of them out, because they are all genetically the same. Sexual reproduction “shuffles the deck” every generation, and this increased variation may help populations survive things like new diseases, climatic shifts, or other environmental changes.
It’s hard to really directly figure out the costs and benefits associated with sexual reproduction. We usually have nothing to compare, because most animals just reproduce sexually. But lucky for me, there are some really weird salamanders around here that break all the reproduction rules. We call them “unisexuals,” because there is only one sex – they are all female. To reproduce, they mate with males of other species of salamanders. Most of the time, despite mating, they actually just reproduce asexually; the dad salamander’s genes do not get included in the offspring. But that’s only most of the time. There are also babies produced that DO have dad’s genome added in. So this means if the mom had two sets of chromosomes (like you and I do), the offspring might end up with three sets – and this somehow works just fine for these salamanders. In fact, the vast majority of them are walking around with three full sets of chromosomes!
 Three salamanders, nine sets of chromosomes
Three salamanders, nine sets of chromosomes
This means that these salamanders are able to reproduce both asexually and sexually, which is very strange for a vertebrate animal. One of the big questions I’m interested in is… WHY? Are there certain times or environmental conditions when it’s better to be asexual, and other conditions where adding dad’s genome is the best bet? So far we don’t have solid answers to these questions. We know that adding the male’s genome is more common at warmer temperatures. We also have some early results suggesting that females are more likely to add the genome if the male is from their same pond, versus a more distant pond. The suggestion that the females may be discriminating against non-local males is intriguing, but we need to do some more experiments to see if these results hold up. Stay tuned for more breeding experiments (and baby salamander pictures) in the spring!
Undergraduate researcher Marisa Hildebrandt will be presenting her research at the Sigma Xi Student Research Conference later this month, and she received a Dean’s Student Travel Award to support her trip. Congratulations, Marisa!
Welcome to the new website of the Greenwald Lab at Eastern Michigan University!